A pedicled skin flap is a piece of tissue transferred from the donor site and inserted into the recipient site while maintaining a continuous attachment to the body (the pedicle). It’s important to understand flap physiology in detail since the creation of any cutaneous flap applies specific stresses to the otherwise normal skin:
- Local tissue trauma,
- Neuro-vascular compromise
The extent to which the skin can survives these stresses is a reflection of:
- Anatomy and physiology of the skin,
- Cutaneous response to the injury.
These principles have led to improved flap survival by: Flap design and Flap delay.
Skin physiology
There are 2 roles of blood supply to skin:
- Nutrition,
- Thermoregulation.
These roles respectively met by two different sets of vascular systems:
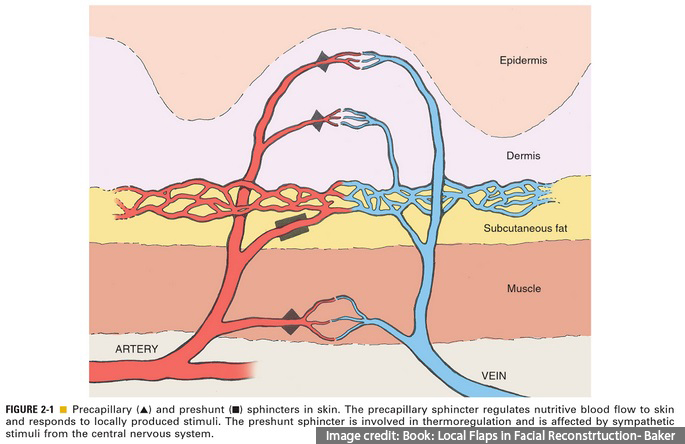
- Nutrient capillary network– in the deeper reticular dermis- precapillary sphincter dilates in response to hypoxemia and increased local metabolic byproduct causing vasodilatation.
- Arteriovenous (AV) shunts in the superficial papillary dermis help in thermoregulation– preshunt sphincter contract to norepinephrine (NE) release by postganglionic sympathetic fibres. With increased body temperature, the NE release decrease. Local release of Acetyl choline by sympathetic nerve fibres can cause vasodilatation by direct fibres or through release of Bradykinin from sweat glands.
Neuro-vascular supply of local skin
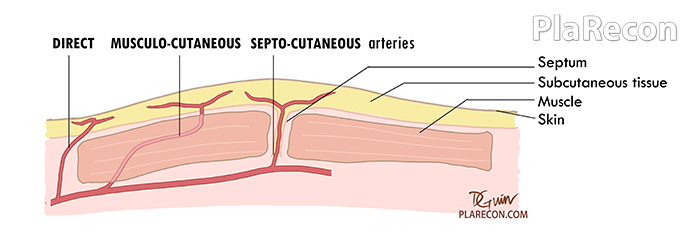
The vessels can be either- 1) Musculo-cutaneous, 2) Direct cutaneous of, 3) Septo- cutaneous.
So, vascular design of a local skin flap based on the vascular anatomy can be either – 1) Random, or 2) Axial-pattern (Arterial)
Acute Physiologic changes after skin flap elevation
A number of changes detrimental to skin survival occur like:
1. Impairment of vascular supply
The survival length of the random part of the flap depends on the physical properties of the supplying vessels (intra- vascular resistance) and the perfusion pressure (Milton).
Fresh flap is both ischemic and viable, depending on the degree of ischemia and the amount of time before recovery of the nutrient blood flow- flap will survive or die (Myers).
Ischemia stimulates neovascularization if the recipient site is favorable: 3-7 days- Angiogenesis- 0.2mm/day- 2-5mm
There is venous (congestion) and lymphatic outflow obstruction (edema).
2. Nerve section
Both cutaneous (loss of sensation) and sympathetic nerves (denervation hyperadrenergic state). Stored catecholamine transmitters (norepinephrine) deplete in 24-48 hours and blood flow increases. In this period-in the critical areas of the random pattern flap, recovery of the nutrient blood flow may be delayed sufficiently to produce additional necrosis.
Severe sympathetic denervation also releases O2 free radicals- inhibit neovascularization.
3. Inflammation (Prostaglandins)
Surgical trauma leads to an inflammatory response.
PGI2 (prostacyclin) levels peak after 7 days while PGE2, PGF2a and TxB2 levels elevate early and return to near-normal in 7 days of flap elevation.
4. Reperfusion (Free radicals)
Once the blood flow returns to the flap after depletion of norepinephrine by 12 hours and reach maximum by 24 hrs- O2 becomes available and a byproduct of reperfusion (free radicals)is produced- Post-ischemic tissue necrosis.
Flap wound healing
Apart from the abovementioned four acute physiological changes, interplay of additional mechanisms (following four cascades) also play equal role in proper flap wound healing and ultimate fate of the flap:
- Clotting
- Compliment
- Kinin
- Plasminogen
Attempts to increase Skin Flap Viability
1. Flap Delay
The use of flap delay has decreased considerably over the last 50 years as:
- flap designs have become more refined and anatomically based
- improved surgical techniques (including microsurgery) have led to more reliably successful dissection and handling of critical structures.
Most frequently now in TRAM flap procedures and head and neck flap cases.
Despite its use as a reliable method for preventing flap necrosis, the mechanisms by which surgical delay acts to improve survival are poorly understood. “Sublethal ischemia” is considered the initiator of the delay phenomenon.
The hypotheses that have been proposed to explain the mechanisms of the delay phenomenon include:
- closing of the arteriovenous shunts,
- conditioning of tissues to survive hypoxia due to ischemia,
- improving blood flow to the flap by vasodilatation of pre-existing vessels.
These hypotheses were based on the findings of circulating catecholamines at the end of the delay period, which cause closure of the A-V shunts, arachidonic acid metabolites that play important roles in the progressive ischemia of the flap and inflammation, and sympathectomy and denervation hypersensitivity during delay procedure, which cause vasodilatation.
Pharmacological Delay
Identification of a critical factor in the delay phenomenon could lead to use of the factor to produce the phenomenon without performing a surgical procedure- so-called “pharmacological delay” (never been reliably demonstrated).
VEGF expressions and bFGF were found to be significantly increased in the tissue with surgical delay due to partial ischaemia – leads to neovascularization in the skin paddle in the late period of surgical delay (3-7 days) by promoting angiogenesis. VEGF is a heparin-binding glycoprotein which is the fundamental regulator in cell signaling of angiogenesis. bFGF also has a strong affinity for the glycosaminoglycan heparin sulfate and is a potent mitogen for proliferation of capillary endothelial cells. VEGF also cause vasodilatation partly through stimulation of nitric oxide synthase in endothelial cells.
Improved flap survival in many models has been observed following manipulation of TGF isoforms, basic fibroblast growth factor (bFGF), PDGF and VEGF.
2. Opening up of ‘Choke vessels’
The ‘choke vessels’, are narrower (reduced-calibre) anastomosing vessels between two adjacent angiosomes that normally act as a barrier in the vasculature but have the capacity to dilate and increase the blood flow.
By opening up/ dilatation of these choke vessels, adjacent angiosome came be incorporated in large flaps extending beyond a single angiosome, hence increasing viability of larger flaps.
If you found this post useful subscribe below for more such articles & subscribe to our YouTube channel for video tutorials. You can also find us on Facebook, Twitter and Instagram.

Tutorials & tips in Plastic Surgery
+ Weekly updates of high-quality webinars!



Very neat summary.